
Nonpolar covalent bonds are a type of bonding where electrons are equally shared between, usually, two of the same nonmetal. This is due to the two atoms having significantly different electronegativities. Polar covalent bonds are a type of bonding where electrons are unequally shared between two different nonmetals. There are actually two types of covalent bonds: polar covalent bonds and nonpolar covalent bonds. Some properties of covalent bonds include low melting points and weak electroconductivity abilities. In the ionic compound NaCl, sodium (Na) loses an electron and obtains a positive charge, while chlorine (Cl) gains an electron and therefore obtains a negative charge.Ĭovalent bonds are formed when atoms share electrons (usually two nonmetals). Some properties of ionic compounds include very strong bonds, solubility in water, and the ability to strongly conduct heat and electricity. The atom that gains an electron will gain a negative charge and is called an anion (usually a nonmetal). The atom that loses an electron will gain a positive charge and is called a cation (usually a metal). Ionic bonds are formed by the transfer of electrons from one atom to another, usually from a metal to a nonmetal. There are two different types of bonds you should be familiar with: ionic bonds and covalent bonds.

□ Want a quick refresh on electronegativity? Check out our study guide on "Periodic Trends." Types of BondsĮlements bond to achieve the lowest possible energy to reach the highest stability. From here, you can try to compare other elements to where fluorine is located on the periodic table. Remember that fluorine is the most electronegative element on the periodic table, with a value of 4.0. This is one of the five essential periodic trends to know and understand for AP Chemistry. Remember, most things in chemistry try to become more stable! ⚖️ Atoms may become ions in order to achieve a more stable electron configuration. This will make more sense once we look into what happens when elements form a bond.Įssentially, ions are charged atoms or molecules that have gained or lost electrons. Here are some examples:Įlements in group one can all bond with chlorine: LiCl, NaCl, KCl, RbCl.Įlements in group two can all bond with O: MgO, CaO, SrO, BaO.īefore getting into bonds, it is also good to memorize the charges of most elements on the periodic table when they bond with another element. Therefore, elements in the same group tend to bond with similar elements and form similar compounds. Remember that the number of valence electrons an atom has affects the way the element bonds with others.
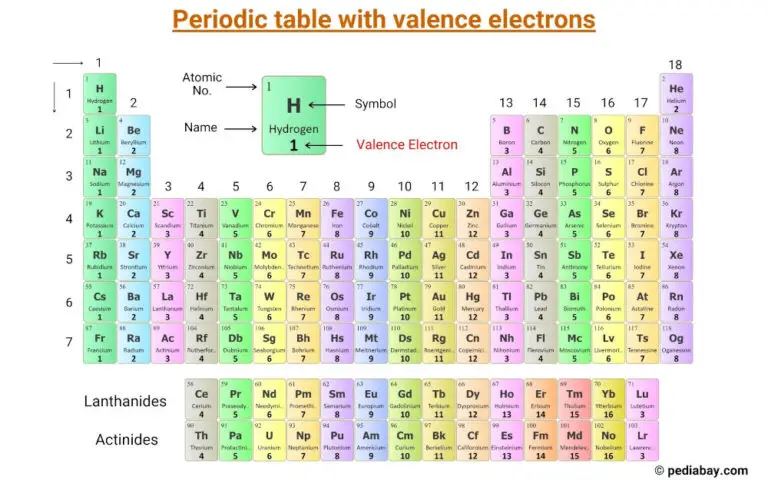
Just by looking at the periodic table, you could tell that oxygen has six valence electrons and carbon has four valence electrons. The AP Chemistry exam is going to primarily focus on the other elements, but it is good to be familiar with several transition metals (like Co, Cu, Zn). The graph above skips over the transition metals because those are much less predictable.

